JCO: 放射免疫结合移植方案治疗DLBCL复发患者无明显获益
2013-04-15 JCO dxy
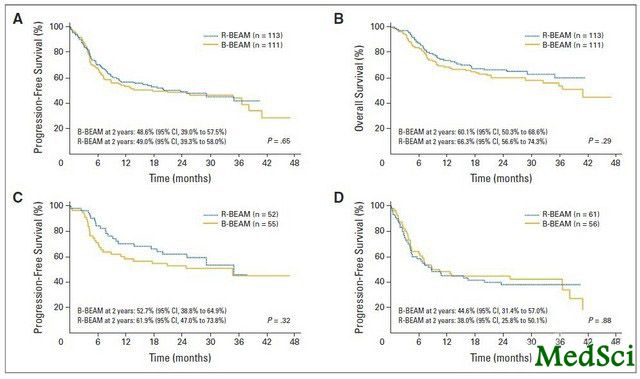
患者OS与PFS结果 在2013年3月11日在线出版的《临床肿瘤学杂志》(Journal of Clinical Oncology)上,美国内布拉斯加大学医学中心的Julie M. Vose等人发表了BMTCTN0401临床试验的III期结果。该项临床试验面向弥漫性大 B 细胞淋巴瘤(DLBCL)复发患者,对以下两种治疗方案进行了对比评价:通过常规剂量的放射免疫疗法结合以化疗为基础的移植治疗方
在2013年3月11日在线出版的《临床肿瘤学杂志》(Journal of Clinical Oncology)上,美国内布拉斯加大学医学中心的Julie M. Vose等人发表了BMTCTN0401临床试验的III期结果。该项临床试验面向弥漫性大 B 细胞淋巴瘤(DLBCL)复发患者,对以下两种治疗方案进行了对比评价:通过常规剂量的放射免疫疗法结合以化疗为基础的移植治疗方案后,继以自体造血干细胞移植治疗;或利妥昔单抗结合相同治疗方案进行治疗;该临床试验编码为NCT00329030。
研究人员对化疗存在持久敏感性或复发性DLBCL患者进行了随机分配,患者接受了碘-131托西莫单抗(第-19天时的剂量为5 mCi,在第-12天时的治疗剂量为0.75 Gy )、卡莫司汀300 mg/m2 (第-6天)、每日两次每次100 mg/m2的依托泊苷(第-5天至第-2天)、每日两次每次100 mg/m2的阿糖胞苷(第-5天至第-2天)、140 mg/m2 美法仑(第-1天; B-BEAM)治疗,或在第-19天至第-12天进行375 mg/m2的利妥昔单抗结合相同的化疗方案治疗(R-BEAM)。
该研究共招募了224例患者,其中113例患者被随机分入R-BEAM组,11例患者分入B-BEAM组。研究人员发现,在研究主要终点,即2年无进展生存率(PFS)方面,R-BEAM为48.6% (95% CI, 38.6% 至57.8%),而B-BEAM组为47.9% (95% CI, 38.2%至57%; P = .94),R-BEAM组患者的2年总生存率(OS)为65.6% (95% CI, 55.3%至74.1%),B-BEAM组为61% (95% CI, 50.9%至69.9%; P = .38)。R-BEAM组患者100天内的治疗相关死亡率为4.1% (95% CI, 0.2%至8.0%),B-BEAM组为4.9% (95% CI, 0.8%至9.0%; P = .97)。此外,与R-BEAM组(0.31)相比,B-BEAM组患者的最大黏膜炎评分较高(0.72; P < .001)。
研究人员最后认为,对于化疗敏感性或复发性DLBCL患者,B-BEAM与R-BEAM治疗方案在2年PFS及OS方面效果类似。另除黏膜炎外,未发现二者在毒性方面存在差异。
与免疫相关的拓展阅读:
- JBC:miR-155对间充质干细胞的免疫调节功能发挥重要调节作用
- JAMA:舌下免疫疗法能用于过敏性鼻炎和哮喘的治疗
- PNAS:陈赛娟白血病治疗药物毛萼乙素在自身免疫病领域研究获进展
- 北京友谊医院院专家走入全球免疫组化质控国际舞台
- Neurology:成人发作型自身免疫性舞蹈症进展迅速
- Nature:食盐或是导致自身免疫性疾病 更多信息请点击:有关免疫更多资讯

Phase III Randomized Study of Rituximab/Carmustine, Etoposide, Cytarabine, and Melphalan (BEAM) Compared With Iodine-131 Tositumomab/BEAM With Autologous Hematopoietic Cell Transplantation for Relapsed Diffuse Large B-Cell Lymphoma: Results From the BMT CTN 0401 Trial.
Abstract
PURPOSEThis clinical trial evaluated standard-dose radioimmunotherapy with a chemotherapy-based transplantation regimen followed by autologous hematopoietic cell transplantation versus rituximab with the same regimen in patients with relapsed diffuse large B-cell lymphoma (DLBCL). PATIENTS AND METHODSPatients with chemotherapy-sensitive persistent or relapsed DLBCL were randomly assigned to receive iodine-131 tositumomab (dosimetric dose of 5 mCi on day -19 and therapeutic dose of 0.75 Gy on day -12), carmustine 300 mg/m2 (day -6), etoposide 100 mg/m2 twice daily (days -5 to -2), cytarabine 100 mg/m2 twice daily (days -5 to -2), and melphalan 140 mg/m2 (day -1; B-BEAM) or rituximab 375 mg/m2 on days -19 and -12 and the same chemotherapy regimen (R-BEAM).ResultsTwo hundred twenty-four patients were enrolled, with 113 patients randomly assigned to R-BEAM and 111 patients assigned to B-BEAM. Two-year progression-free survival (PFS) rates, the primary end point, were 48.6% (95% CI, 38.6% to 57.8%) for R-BEAM and 47.9% (95% CI, 38.2% to 57%; P = .94) for B-BEAM, and the 2-year overall survival (OS) rates were 65.6% (95% CI, 55.3% to 74.1%) for R-BEAM and 61% (95% CI, 50.9% to 69.9%; P = .38) for B-BEAM. The 100-day treatment-related mortality rates were 4.1% (95% CI, 0.2% to 8.0%) for R-BEAM and 4.9% (95% CI, 0.8% to 9.0%; P = .97) for B-BEAM. The maximum mucositis score was higher in the B-BEAM arm (0.72) compared with the R-BEAM arm (0.31; P < .001). CONCLUSIONThe B-BEAM and R-BEAM regimens produced similar 2-year PFS and OS rates for patients with chemotherapy-sensitive relapsed DLBCL. No differences in toxicities other than mucositis were noted.
本网站所有内容来源注明为“梅斯医学”或“MedSci原创”的文字、图片和音视频资料,版权均属于梅斯医学所有。非经授权,任何媒体、网站或个人不得转载,授权转载时须注明来源为“梅斯医学”。其它来源的文章系转载文章,或“梅斯号”自媒体发布的文章,仅系出于传递更多信息之目的,本站仅负责审核内容合规,其内容不代表本站立场,本站不负责内容的准确性和版权。如果存在侵权、或不希望被转载的媒体或个人可与我们联系,我们将立即进行删除处理。
在此留言







#JCO#
17
#LBCL#
35
#DLBCL#
28