2019年阿尔茨海默病十大临床研究进展
2020-01-05 神经科学临床和基础 神经科学临床和基础
1、NEJM:BACE-1抑制剂Verubecestat治疗前驱性AD痴呆研究失败 1454名患者参与研究,其中12mg剂量组485人,40mg剂量组484人,安慰剂组485人。12mg剂量组、40mg剂量组以及安慰剂组104周CDR-SB得分变化分别为1.65、2.01以及1.58,高剂量组患者治疗后症状加重,各组患者每100随访年确诊AD病例分别为24.5、25.5以及19.3例,相比于
1、NEJM:BACE-1抑制剂Verubecestat治疗前驱性AD痴呆研究失败
1454名患者参与研究,其中12mg剂量组485人,40mg剂量组484人,安慰剂组485人。12mg剂量组、40mg剂量组以及安慰剂组104周CDR-SB得分变化分别为1.65、2.01以及1.58,高剂量组患者治疗后症状加重,各组患者每100随访年确诊AD病例分别为24.5、25.5以及19.3例,相比于安慰剂, 40mg剂量组AD风险为1.38。Verubecestat治疗后导致的不良事件增加。
参考文献:
Eganet al (2019). Randomized Trial of Verubecestat for Prodromal Alzheimer'sDisease. N Engl J Med. 2019 Apr 11;380(15):1408-1420.
2. 对于高血压患者而言,服用降压药确实可以降低罹患阿尔茨海默病的风险
JAMA:控制血压能降低阿尔茨海默症风险?一项超过9000人的收缩压干预试验(Systolic Blood Pressure Intervention Trial,SPRINT)的分支试验给出了一份数据:科学家们发现,试验中将血压降至120(收缩压)的人患轻度认知障碍的可能性降低了19%。
参考文献:
Dinget al (2019)—Antihypertensive medications and risk for incident dementiaand Alzheimer's disease: a meta-analysis of individual participant data fromprospective cohort studies. Lancet Neurol. 2020 Jan;19(1):61-70.
Jeff D. Williamson, et al. Effect of Intensive vs Standard Blood Pressure Control on Probable Dementia: A Randomized Clinical Trial. JAMA. Jan 2019.
3.Nat Med:罕见基因突变或能预防阿尔茨海默病
一名女性的大脑淀粉样蛋白病理改变程度较高,这也是阿尔茨海默病的一大特征。但是,这名女性并没有出现与该病相关的症状。美国麻省总医院、麻省眼耳医院Schepens眼科研究所与哈佛大学医学院眼科系以及Banner阿尔茨海默病研究所的科学家们,一同研究了她的基因组,发现除致病突变外,她还携带一个APOE基因的罕见突变,名为“Christchurch”。
研究人员认为,这一突变可能抵消了早老素1突变的有害作用,导致该女性脑内的淀粉样病理改变程度虽高,但却并没有患上该疾病。这名女性的认知健康一直维持到70多岁。经过多次实验,团队认为这种突变可能破坏了APOE与阿尔茨海默病有关糖类的结合,从而产生了保护作用。
参考文献:
Arboleda-Velasquez et al (2019). Resistance to autosomaldominant Alzheimer's disease in an APOE3 Christchurch homozygote: a casereport. Nat Med. 2019 Nov;25(11):1680-1683.
4. Lancet Neurol—全球疾病负担研究组发布1990-2016年间全球阿尔茨海默病和其他痴呆的疾病负担数据
英文摘要:
BACKGROUND:
Thenumber of individuals living with dementia is increasing, negatively affectingfamilies, communities, and health-care systems around the world. A successfulresponse to these challenges requires an accurate understanding of the dementiadisease burden. We aimed to present the first detailed analysis of the globalprevalence, mortality, and overall burden of dementia as captured by the GlobalBurden of Diseases, Injuries, and Risk Factors (GBD) Study 2016, and highlightthe most important messages for clinicians and neurologists.
METHODS:
GBD2016 obtained data on dementia from vital registration systems, publishedscientific literature and surveys, and data from health-service encounters ondeaths, excess mortality, prevalence, and incidence from 195 countries andterritories from 1990 to 2016, through systematic review and additionaldata-seeking efforts. To correct for differences in cause of death codingacross time and locations, we modelled mortality due to dementia usingprevalence data and estimates of excess mortality derived from countries thatwere most likely to code deaths to dementia relative to prevalence. Data wereanalysed by standardised methods to estimate deaths, prevalence, years of lifelost (YLLs), years of life lived with disability (YLDs), anddisability-adjusted life-years (DALYs; computed as the sum of YLLs and YLDs),and the fractions of these metrics that were attributable to four risk factorsthat met GBD criteria for assessment (high body-mass index [BMI], high fastingplasma glucose, smoking, and a diet high in sugar-sweetened beverages).
FINDINGS:
In2016, the global number of individuals who lived with dementia was 43·8 million(95% uncertainty interval [UI] 37·8-51·0), increased from 20.2 million(17·4-23·5) in 1990. This increase of 117% (95% UI 114-121) contrasted with aminor increase in age-standardised prevalence of 1·7% (1·0-2·4), from 701 cases(95% UI 602-815) per 100 000 population in 1990 to 712 cases (614-828) per100 000 population in 2016. More women than men had dementia in 2016 (27·0million, 95% UI 23·3-31·4, vs 16.8 million, 14.4-19.6), and dementia was thefifth leading cause of death globally, accounting for 2·4 million (95% UI2·1-2·8) deaths. Overall, 28·8 million (95% UI 24·5-34·0) DALYs were attributedto dementia; 6·4 million (95% UI 3·4-10·5) of these could be attributed to themodifiable GBD risk factors of high BMI, high fasting plasma glucose, smoking,and a high intake of sugar-sweetened beverages.
INTERPRETATION:
Theglobal number of people living with dementia more than doubled from 1990 to2016, mainly due to increases in population ageing and growth. Althoughdifferences in coding for causes of death and the heterogeneity incase-ascertainment methods constitute major challenges to the estimation of theburden of dementia, future analyses should improve on the methods for thecorrection of these biases. Until breakthroughs are made in prevention orcurative treatment, dementia will constitute an increasing challenge tohealth-care systems worldwide.
参考文献:
GBD2016 Dementia Collaborators. Global, regional, and national burden ofAlzheimer's disease and other dementias, 1990-2016: a systematic analysis forthe Global Burden of Disease Study 2016. Lancet Neurol. 2019 Jan;18(1):88-106.
5. Nature medicine和Cell stem cell—今年最大的学术争议之一:阿尔茨海默病中成年海马神经再生是否持续存在?
英文摘要1:
The hippocampus is one of the most affected areas in Alzheimer's disease (AD)1. Moreover, thisstructure hosts one of the most unique phenomena of the adult mammalian brain,namely, the addition of new neurons throughout life2. This process,called adult hippocampal neurogenesis (AHN), confers an unparalleled degree ofplasticity to the entire hippocampal circuitry3,4. Nonetheless,direct evidence of AHN in humans has remained elusive. Thus, determiningwhether new neurons are continuously incorporated into the human dentate gyrus(DG) during physiological and pathological aging is a crucial question withoutstanding therapeutic potential. By combining human brain samples obtainedunder tightly controlled conditions and state-of-the-art tissue processingmethods, we identified thousands of immature neurons in the DG ofneurologically healthy human subjects up to the ninth decade of life. Theseneurons exhibited variable degrees of maturation along differentiation stagesof AHN. In sharp contrast, the number and maturation of these neuronsprogressively declined as AD advanced. These results demonstrate thepersistence of AHN during both physiological and pathological aging in humansand provide evidence for impaired neurogenesis as a potentially relevantmechanism underlying memory deficits in AD that might be amenable to noveltherapeutic strategies.
英文摘要2:
Whetherhippocampal neurogenesis persists throughout life in the human brain is notfully resolved. Here, we demonstrate that hippocampal neurogenesis ispersistent through the tenth decade of life and is detectable in patients withmild cognitive impairments and Alzheimer's disease.In a cohort of 18 participants with a mean age of 90.6 years, Nestin+Sox2+neural progenitor cells (NPCs) and DCX+ neuroblasts and immatureneurons were detected, but their numbers greatly varied between participants.Nestin+ cells localize in the anterior hippocampus, and NPCs,neuroblasts, and immature neurons are evenly distributed along the anterior toposterior axis. The number of DCX+PCNA+ cells is reducedin mild cognitive impairments, and higher numbers of neuroblasts are associatedwith better cognitive status. The number of DCX+PCNA+cells correlates with functional interactions between presynaptic SNAREproteins. Our results suggest that hippocampal neurogenesis persists in theaged and diseased human brain and that it is possibly associated withcognition.
参考文献:
1.Moreno-Jiménez et al (2019). Adult hippocampal neurogenesis is abundant inneurologically healthy subjects and drops sharply in patients with Alzheimer'sdisease. Nat Med. 2019 Apr;25(4):554-560.
2.Tobin et al (2019). Human Hippocampal Neurogenesis Persists in Aged Adults andAlzheimer's Disease Patients. Cell Stem Cell. 2019 Jun 6;24(6):974-982.e3.
6. Lancet Neurol—科学家构建了一个可以高效预测MCI进展为痴呆的生物标志物模型
英文摘要:
BACKGROUND:
Biomarker-basedrisk predictions of dementia in people with mild cognitive impairment arehighly relevant for care planning and to select patients for treatment whendisease-modifying drugs become available. We aimed to establish robustprediction models of disease progression in people at risk of dementia.
METHODS:
Inthis modelling study, we included people with mild cognitive impairment (MCI)from single-centre and multicentre cohorts in Europe and North America: theEuropean Medical Information Framework for Alzheimer's Disease (EMIF-AD;n=883), Alzheimer's Disease Neuroimaging Initiative (ADNI; n=829), AmsterdamDementia Cohort (ADC; n=666), and the Swedish BioFINDER study (n=233).Inclusion criteria were a baseline diagnosis of MCI, at least 6 months offollow-up, and availability of a baseline Mini-Mental State Examination (MMSE)and MRI or CSF biomarker assessment. The primary endpoint was clinicalprogression to any type of dementia. We evaluated performance of previouslydeveloped risk prediction models-a demographics model, a hippocampal volumemodel, and a CSF biomarkers model-by evaluating them across cohorts,incorporating different biomarker measurement methods, and determiningprognostic performance with Harrell's C statistic. We then updated the modelsby re-estimating parameters with and without centre-specific effects andevaluated model calibration by comparing observed and expected survival.Finally, we constructed a model combining markers for amyloid deposition,tauopathy, and neurodegeneration (ATN), in accordance with the NationalInstitute on Aging and Alzheimer's Association research framework.
FINDINGS:
Weincluded all 2611 individuals with MCI in the four cohorts, 1007 (39%) of whomprogressed to dementia. The validated demographics model (Harrell's C 0·62, 95%CI 0·59-0·65), validated hippocampal volume model (0·67, 0·62-0·72), andupdated CSF biomarkers model (0·72, 0·68-0·74) had adequate prognosticperformance across cohorts and were well calibrated. The newly constructed ATNmodel had the highest performance (0·74, 0·71-0·76).
INTERPRETATION:
Wegenerated risk models that are robust across cohorts, which adds to theirpotential clinical applicability. The models could aid clinicians in theinterpretation of CSF biomarker and hippocampal volume results in individualswith MCI, and help research and clinical settings to prepare for a future ofprecision medicine in Alzheimer's disease. Future research should focus on theclinical utility of the models, particularly if their use affects participants'understanding, emotional wellbeing, and behaviour.
FUNDING:
ZonMW-Memorabel.
参考文献:
Van et al (2019). Biomarker-based prognosis for peoplewith mild cognitive impairment (ABIDE): a modelling study. Lancet Neurol. 2019Nov;18(11):1034-1044.
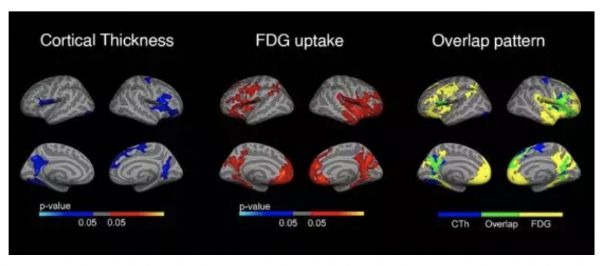
7. JAMA—研究发现ATN模型可以有助于预测非痴呆老年人群的记忆力下降
英文摘要:
Importance:
ANational Institute on Aging and Alzheimer's Association workgroup proposed aresearch framework for Alzheimer disease in which biomarker classification ofresearch participants is labeled AT(N) for amyloid, tau, and neurodegenerationbiomarkers.
Objective:
Todetermine the associations between AT(N) biomarker profiles and memory declinein a population-based cohort of individuals without dementia age 60 years orolder, and to determine whether biomarkers provide incremental prognostic valuebeyond more readily available clinical and genetic information.
Design,Setting, and Participants:
Population-basedcohort study of cognitive aging in Olmsted County, Minnesota, that included 480nondemented Mayo Clinic Study of Aging participants who had a clinical evaluationand amyloid positron emission tomography (PET) (A), tau PET (T), and magneticresonance imaging (MRI) cortical thickness (N) measures between April 16, 2015,and November 1, 2017, and at least 1 clinical evaluation follow-up by November12, 2018.
Exposures:
Age,sex, education, cardiovascular and metabolic conditions score, APOE genotype,and AT(N) biomarker profiles. Each of A, T, or (N) can be abnormal (+) ornormal (-), resulting in 8 AT(N) profiles.
MainOutcomes and Measures:
Primaryoutcome was a composite memory score measured longitudinally at 15-monthintervals. Analyses measured the associations between predictor variables andthe memory score, and whether AT(N) biomarker profiles significantly improvedprediction of memory z score rates of change beyond a model with clinical andgenetic variables only.
Results:
Participantswere followed up for a median of 4.8 years (interquartile range [IQR], 3.8-5.1)and 44% were women (211/480). Median (IQR) ages ranged from 67 years (65-73) inthe A-T-(N)- group to 83 years (76-87) in the A+T+(N)+ group. Of theparticipants, 92% (441/480) were cognitively unimpaired but the A+T+(N)+ grouphad the largest proportion of mild cognitive impairment (30%). AT(N) biomarkersimproved the prediction of memory performance over a clinical model from an R2of 0.26 to 0.31 (P < .001). Memory declined fastest in the A+T+(N)+,A+T+(N)-, and A+T-(N)+ groups compared with the other 5 AT(N) groups(P = .002). Estimated rates of decline in the 3 fastest declining groups were-0.13 (95% CI, -0.17 to -0.09), -0.10 (95% CI, -0.16 to -0.05), and -0.10 (95%CI, -0.13 to -0.06) z score units per year, respectively, for an 85-year-oldAPOE ε4 noncarrier.
Conclusionsand Relevance:
Amongolder persons without baseline dementia followed for a median of 4.8 years, aprediction model that included amyloid PET, tau PET, and MRI cortical thicknessresulted in a small but statistically significant improvement in predictingmemory decline over a model with more readily available clinical and geneticvariables. The clinical importance of this difference is uncertain.
参考文献:
Jacket al (2019). Associations of Amyloid, Tau, and Neurodegeneration BiomarkerProfiles With Rates of Memory Decline Among Individuals Without Dementia. JAMA.2019 Jun 18;321(23):2316-2325.
8. Nature medicine—血清神经丝轻链水平可以预测无症状阿尔次海默病进展为痴呆
英文摘要:
Neurofilamentlight chain (NfL) is a promising fluid biomarker of disease progression forvarious cerebral proteopathies. Here we leverage the unique characteristics ofthe Dominantly Inherited Alzheimer Network and ultrasensitive immunoassaytechnology to demonstrate that NfL levels in the cerebrospinal fluid (n = 187)and serum (n = 405) are correlated with one another and are elevated at thepresymptomatic stages of familial Alzheimer's disease. Longitudinal,within-person analysis of serum NfL dynamics (n = 196) confirmed this elevationand further revealed that the rate of change of serum NfL could discriminatemutation carriers from non-mutation carriers almost a decade earlier thancross-sectional absolute NfL levels (that is, 16.2 versus 6.8 years before theestimated symptom onset). Serum NfL rate of change peaked in participantsconverting from the presymptomatic to the symptomatic stage and was associatedwith cortical thinning assessed by magnetic resonance imaging, but less so withamyloid-β deposition or glucose metabolism (assessed by positron emissiontomography). Serum NfL was predictive for both the rate of cortical thinningand cognitive changes assessed by the Mini-Mental State Examination and LogicalMemory test. Thus, NfL dynamics in serum predict disease progression and brainneurodegeneration at the early presymptomatic stages of familial Alzheimer's disease,which supports its potential utility as a clinically useful biomarker.
参考文献:
Preischeet al (2019). Serum neurofilament dynamics predicts neurodegeneration andclinical progression in presymptomatic Alzheimer's disease. Nat Med. 2019Feb;25(2):277-283.
9. Lancet Neurol—脆弱程度或许可以用来解释阿尔茨海默病临床和病理不匹配的矛盾
英文摘要:
BACKGROUND:
Somepeople with substantial Alzheimer's disease pathology at autopsy had shown fewcharacteristic clinical symptoms or signs of the disease, whereas others withlittle Alzheimer's disease pathology have been diagnosed with Alzheimer'sdementia. We aimed to examine whether frailty, which is associated with bothage and dementia, moderates the relationship between Alzheimer's diseasepathology and Alzheimer's dementia.
METHODS:
Wedid a cross-sectional analysis of data from participants of the Rush Memory andAging Project, a clinical-pathological cohort study of older adults (older than59 years) without known dementia at baseline, living in Illinois, USA. Participantsin the cohort study underwent annual neuropsychological and clinicalevaluations. In the present cross-sectional analysis, we included thoseparticipants who did not have any form of dementia or who had Alzheimer'sdementia at the time of their last clinical assessment and who had died and forwhom complete autopsy data were available. Alzheimer's disease pathology wasquantified by a summary measure of neurofibrillary tangles and neuritic anddiffuse plaques. Clinical diagnosis of Alzheimer's dementia was based onclinician consensus. Frailty was operationalised retrospectively using healthvariable information obtained at each clincial evaluation using the deficitaccumulation approach (41-item frailty index). Logistic regression andmoderation modelling were used to assess relationships between Alzheimer'sdisease pathology, frailty, and Alzheimer's dementia. All analyses wereadjusted for age, sex, and education.
FINDINGS:
Upto data cutoff (Jan 20, 2017), we included 456 participants (mean age at death89·7 years [SD 6·1]; 316 [69%] women). 242 (53%) had a diagnosis of possible orprobable Alzheimer's dementia at their last clinical assessment. Frailty (oddsratio 1·76, 95% CI 1·54-2·02; p<0·0001) and Alzheimer's disease pathology(4·81, 3·31-7·01; p<0·0001) were independently associated with Alzheimer'sdementia, after adjusting for age, sex, and education. When frailty was addedto the model for the relationship between Alzheimer's disease pathology andAlzheimer's dementia, model fit improved (p<0·0001). There was a significantinteraction between frailty and Alzheimer's disease pathology (odds ratio 0·73,95% CI 0·57-0·94; pinteraction=0·015). People with an increased frailty scorehad a weakened direct link between Alzheimer's disease pathology andAlzheimer's dementia; that is, people with a low amount of frailty were betterable to tolerate Alzheimer's disease pathology, whereas those with higheramounts of frailty were more likely both to have more Alzheimer's diseasepathology and for it to be expressed as dementia.
INTERPRETATION:
Thedegree of frailty among people of the same age modifies the association betweenAlzheimer's disease pathology and Alzheimer's dementia. That frailty is relatedto both odds of Alzheimer's dementia and disease expression has implicationsfor clinical management, since individuals with even a low level of Alzheimer'sdisease pathology might be at risk for dementia if they have high amounts offrailty. Further research should assess how frailty and cognition change overtime to better elucidate this complex relationship.
参考文献:
Wallaceet al (2019). Investigation of frailty as a moderator of the relationshipbetween neuropathology and dementia in Alzheimer's disease: a cross-sectionalanalysis of data from the Rush Memory and Aging Project. Lancet Neurol. 2019Feb;18(2):177-18.
10. N Engl J Med—穹窿区深部脑刺激可以诱导阿尔茨海默病患者的记忆回想
英文摘要:
In atrial of stimulation of the fornix and subcallosal regions of the hippocampusinvolving 42 patients with Alzheimer’s disease, 20 patients reported vividmemory flashbacks. The robustness and complexity of the memories increased withincreasing voltage applied to the stimulating electrodes.
参考文献:
Deebet al (2019). Fornix-Region Deep Brain Stimulation-Induced Memory Flashbacks in Alzheimer's Disease. N Engl J Med. 2019 Aug 22;381(8):783-785.
本网站所有内容来源注明为“梅斯医学”或“MedSci原创”的文字、图片和音视频资料,版权均属于梅斯医学所有。非经授权,任何媒体、网站或个人不得转载,授权转载时须注明来源为“梅斯医学”。其它来源的文章系转载文章,或“梅斯号”自媒体发布的文章,仅系出于传递更多信息之目的,本站仅负责审核内容合规,其内容不代表本站立场,本站不负责内容的准确性和版权。如果存在侵权、或不希望被转载的媒体或个人可与我们联系,我们将立即进行删除处理。
在此留言













#阿尔茨#
44
#阿尔茨海默病#也即#老年性痴呆#,目前的药物研究,大多是失败的
242
#阿尔茨海#
37
#研究进展#
38
#阿尔茨海默#
45