AHA 2019丨COLCOT试验:COLchicine心血管结局试验
2019-11-17 MedFocus POCKET AHA2019
2019年11月16日,一年一度的美国心脏协会科学年会(AHA)在美国宾夕法尼亚州费城隆重启幕,来自世界各地的15000多名顶尖医生、科学家、心脏病专家、保健专业人员和其他人士参加本次会议,讨论心血管科学和医学的未来。来自加拿大蒙特利尔心脏研究所的Jean-Claude Tardif教授在第一场LBS专场中为大家揭晓了COLCOT试验的最新研究结果,COLCOT试验的结果将确定,用小剂量消炎药秋水
2019年11月16日,一年一度的美国心脏协会科学年会(AHA)在美国宾夕法尼亚州费城隆重启幕,来自世界各地的15000多名顶尖医生、科学家、心脏病专家、保健专业人员和其他人士参加本次会议,讨论心血管科学和医学的未来。来自加拿大蒙特利尔心脏研究所的Jean-Claude Tardif教授在第一场LBS专场中为大家揭晓了COLCOT试验的最新研究结果,COLCOT试验的结果将确定,用小剂量消炎药秋水仙碱治疗患者是否能降低他汀类药物治疗以外的缺血性事件的发生率。
Abstract
AHA 2019|COLCOT试验
Introduction:
Multiple lines of evidence have highlighted the important role of inflammation in the pathogenesis of atherosclerosis and its complications. Colchicine is an orally administered, potent anti-inflammatory drug clinically indicated for the treatment of gout and pericarditis.
多种证据表明炎症在动脉粥样硬化及其并发症的发病机制中起着重要作用。秋水仙碱是一种有效的口服抗炎药,临床上用于治疗痛风和心包炎。
Hypothesis:
We hypothesized that colchicine will reduce cardiovascular (CV) events in patients with a recent myocardial infarction (MI).
Methods:
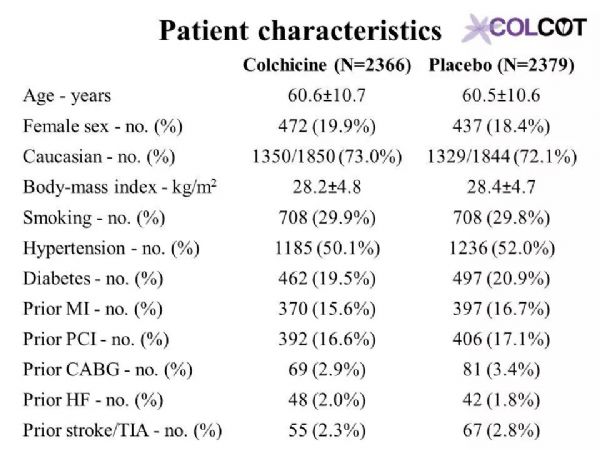
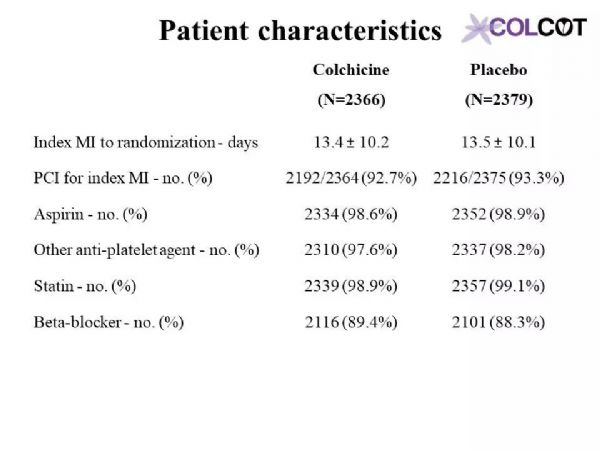
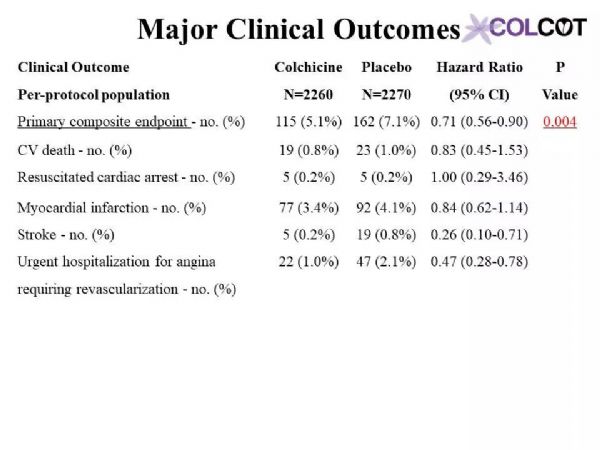
The primary objective of COLCOT is to determine if long-term treatment with colchicine 0.5 mg/day will reduce major CV events in patients with a recent MI. The secondary objective is to determine the safety and tolerability of long-term treatment with low-dose colchicine. COLCOT is a randomized, double-blind, placebo-controlled, multi-national clinical trial. A total of 167 clinical sites participated in the trial. Patients were randomly assigned to receive either colchicine (0.5 mg/day) or placebo (1:1 ratio) and followed until at least 301 primary CV events occurred. To enter the trial, adult men and women had to have suffered a MI within the last 30 days and have completed any planned percutaneous revascularization procedures. Follow-up visits occurred at 1 and 3 months following randomization and every ≈3 months thereafter. Patients also received standard medical care including intensive use of statins. The primary study endpoint is the time from randomization to the first event of CV mortality, resuscitated cardiac arrest, non-fatal MI, non-fatal stroke, or urgent hospitalization for angina requiring coronary revascularization. All suspected CV endpoints were adjudicated by an independent and blinded clinical endpoint committee. COLCOT is an event-driven trial with main analyses conducted on an intention-to-treat basis. The sample size calculation was based on the primary endpoint and a hazard ratio of 0.724. Using a two-sided test at the 0.05 significance level, the trial had 80% power if it continued until 301 positively adjudicated primary events occurred in the combined treatment groups. The total number of patients to randomize, 4500, was chosen so that the expected number of adjudicated events after 24 months of follow-up is 301. It assumed an event rate of 7% in the placebo group at 24 months, an 18-month recruitment period, a 24-month minimum follow-up and a 1% yearly lost to follow-up and withdrawal of consent rate.
COLCOT研究的主要目的是确定长期使用0.5毫克/天的秋水仙碱治疗是否能减少心肌梗死患者的主要心血管事件。次要目的是确定长期的小剂量秋水仙碱治疗的安全性和耐受性。COLCOT是一项随机、双盲、安慰剂对照、多国参与的临床试验。共有167个临床中心参与了试验。患者被随机分配接受秋水仙碱(0.5毫克/天)或安慰剂(1:1比率)治疗,且至少需随访301例主要心血管事件。为了进入试验,成年男女必须在过去30天内经历心肌梗死,并完成任意计划的经皮血运重建手术。随机分组后第1个月和第3个月进行随访,此后每≈3个月进行一次随访。患者还接受了标准的医疗护理,包括强化使用他汀类药物。主要研究终点是从随机化后发生的心血管死亡、复苏性心脏骤停、非致命性心肌梗死、非致命性卒中或因心绞痛需要冠状动脉血运重建而紧急住院的时间。所有可疑的心血管终点都由一个独立的临床终点委员会判定。COLCOT是事件驱动型试验,主要分析是在意向治疗的基础上进行的。样本量计算基于主要终点和0.724的危险比。采用双侧0.05显着性水平,如果试验持续到联合治疗组发生301例阳性判定的原发性事件,则试验有80%的效能。随机选择4500名患者,以便在24个月的随访后,预期的判定事件数为301。假设安慰剂组在24个月发生率为7%、同时18个月的招募期、24个月的最低随访期和每年1%的失访率和退出率。
Results:
A total of 4745 patients were enrolled in COLCOT.
共有4745名患者参加了COLCOT研究。
Limitations:
The duration of follow-up was relatively short at approximately 23 months. The risks and benefits of longer-term treatment with colchicine were not evaluated. Although the inclusion of 4745 patients was sufficient to demonstrate a significant benefit on the primary composite efficacy endpoint, a larger trial could have allowed a better assessment of individual endpoints and subgroups and the risks associated with colchicine.
随访时间较短,约23个月。长期使用秋水仙碱治疗的风险和益处没有评估。尽管纳入4745名患者足以证明在主要复合疗效终点有显着益处,但更大规模的试验可以更好地评估个别终点和亚组以及秋水仙碱相关的风险。
Conclusion:
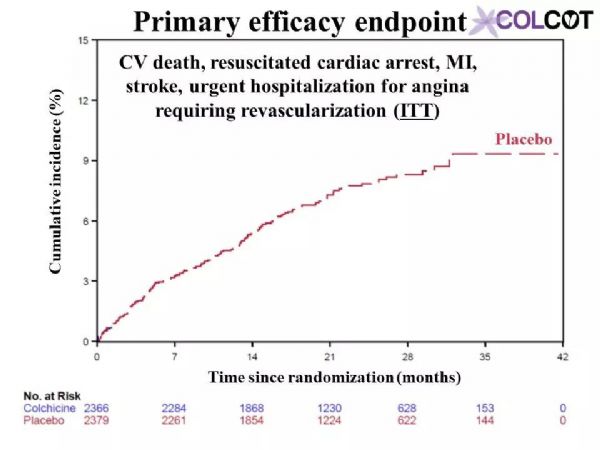
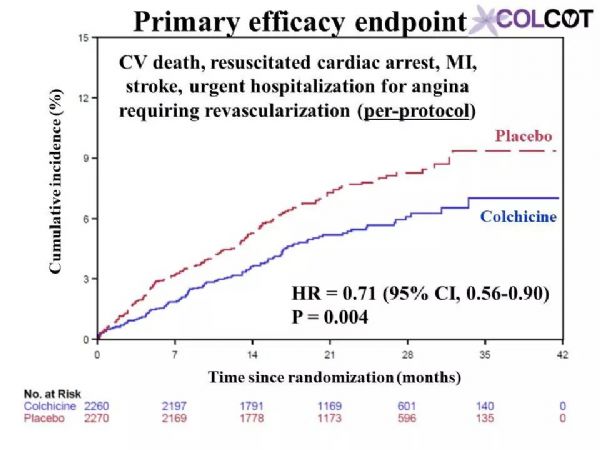
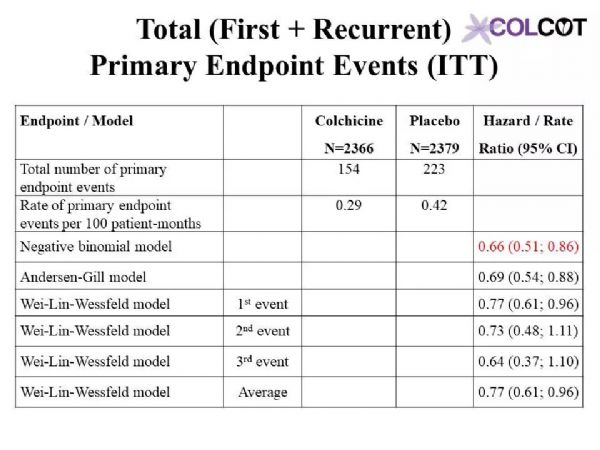
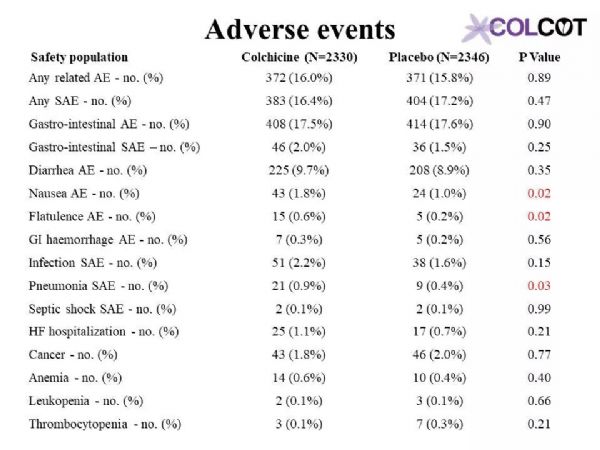
Colchicine 0.5 mg/day significantly reduces the risk of first and total ischemic cardiovascular events by 23% and 34% respectively compared to placebo in patients with a recent myocardial infarction.Rates of adverse effects were low, including a small increase in pneumonias (0.9 vs. 0.4%) but no significant increase in diarrhea with colchicine, on background therapy with aspirin, a 2nd antiplatelet agent and a statin in 99, 98 and 99% of patients.The COLCOT results apply to patients who have recently suffered a myocardial infarction. Further research is needed to assess the benefits of colchicine in other high-risk patients.
与安慰剂组相比,0.5mg秋水仙碱治疗组可显着降低近期心肌梗死患者首次和总缺血性心血管事件的风险,分别降低23%和34%。99%、98%和99%的患者分别有阿司匹林、第二抗血小板药和他汀类药物的用药史,不良反应发生率较低,包括肺炎轻微增加(0.9%对0.4%),但秋水仙碱腹泻无明显增加。COLCOT结果适用于最近发生过心肌梗死的患者。还需要进一步的研究来评估秋水仙碱对其他高危患者的收益。
Slides
AHA 2019|COLCOT试验
POCKET AHA2019
Immediately
由中国心血管健康联盟和心关注共同发起的POCKET AHA2019项目将依托医望医学提供的人工智能学术跟踪系统,为全国广大心血管医师带来及时全面的报道。
项目得到了医师报、365医学网、国际循环、华迈医学、MedFocus等媒体的大力支持。
小提示:本篇资讯需要登录阅读,点击跳转登录
版权声明:
本网站所有内容来源注明为“梅斯医学”或“MedSci原创”的文字、图片和音视频资料,版权均属于梅斯医学所有。非经授权,任何媒体、网站或个人不得转载,授权转载时须注明来源为“梅斯医学”。其它来源的文章系转载文章,或“梅斯号”自媒体发布的文章,仅系出于传递更多信息之目的,本站仅负责审核内容合规,其内容不代表本站立场,本站不负责内容的准确性和版权。如果存在侵权、或不希望被转载的媒体或个人可与我们联系,我们将立即进行删除处理。
在此留言
本网站所有内容来源注明为“梅斯医学”或“MedSci原创”的文字、图片和音视频资料,版权均属于梅斯医学所有。非经授权,任何媒体、网站或个人不得转载,授权转载时须注明来源为“梅斯医学”。其它来源的文章系转载文章,或“梅斯号”自媒体发布的文章,仅系出于传递更多信息之目的,本站仅负责审核内容合规,其内容不代表本站立场,本站不负责内容的准确性和版权。如果存在侵权、或不希望被转载的媒体或个人可与我们联系,我们将立即进行删除处理。
在此留言

















#CIN#
50
#COLCOT#
46
#AHA#
44
#心血管结局#
50
#COLchicine#
37