读书报告 | 阿巴西普联合剂鲁索替尼治疗免疫相关心肌炎
2024-01-07 iCombo iCombo 发表于陕西省
来自法国索邦大学的教授团队,基于心肌炎发病机制调整治疗策略,给与患者基于CD86 RO监测的高剂量阿巴西普、JAK2抑制剂鲁索替尼及系统性筛查和早期呼吸机支持治疗。
导读
免疫检查点抑制剂(immune-checkpoint inhibitors,ICIs)在提高恶性肿瘤治疗疗效同时亦可引发免疫检查点抑制剂相关心肌炎等严重不良事件,虽然发生率只有约1%,但死亡率可达30%-50%,其中导致患者死亡的主要原因包括室性心率失常、心衰、呼吸肌衰竭等。目前针对免疫检查点抑制剂相关心肌炎主要治疗方法为皮质类固醇及低剂量CTLA-4融合蛋白阿巴西普(Abatacept)等,但治疗效果欠佳。现来自法国索邦大学的Yves ALLENBACH教授团队,基于心肌炎发病机制调整治疗策略,给与患者基于CD86 RO监测的高剂量阿巴西普、JAK2抑制剂鲁索替尼(Ruxolitinib)及系统性筛查和早期呼吸机支持治疗,从而明显降低了ICI心肌炎的死亡率至3%,但此用药组合及合理剂量仍需在未来临床研究中进一步评估(Cancer Discov. 2023 Feb 23;CD-22-1180. doi: 10.1158/2159-8290.CD-22-1180)。
研究背景
Immune‐checkpoint inhibitors restore T‐cell‐mediated host immunity against tumors, but they may also induce immune related adverse events (irAE) including ICI‐myocarditis which occurs infrequently (~1%) but carries a 30‐50% mortality rate.
The main causes of death attributed to ICI‐myotoxicities are life‐threatening ventricular arrhythmias, cardiogenic shock or hypercapnic respiratory muscle failure.
Current treatment of ICI‐myocarditis includes empiric corticosteroids, which are not universally effective.
Abatacept is effective in attenuating myocardial inflammation In pre‐clinical models Preliminary case reports.
Preliminary reports suggest abatacept clinical efficacy can be enhanced by addition of the Janus kinase (JAK) inhibitor ruxolitinib.
研究方法
Participants:
1、 prospective single‐center cohort, between 10/05/2018 and 08/18/2021
2、cardio‐oncology unit (Pitié‐Salpétrière, Paris, France)
3、ICI‐ related myotoxicity was confirmed by endomyocardial or muscle biopsies in all patients.
4、Cardiac involvement was evaluated by cardiac MRI, endomyocardial biopsy, coronary angiography, echocardiography, electrocardiography and Troponin‐T monitoring.
5、Skeletal muscle involvement was evaluated by peripheral muscle and diaphragmatic MRI, muscular biopsy, electromyogram, diaphragmatic echography and pulmonary functional test specifically seeking for respiratory muscle dysfunction.
Therapeutic strategy :
Q1(2018.5-2020.3)
1、High dose boluses of corticosteroids regardless of symptoms and severity grade.
2、In severe cases(grade≥3):plasmapheresis, mycophenolate‐mofetil and abatacept (~10mg/kg every 2 weeks).
Q2(2020.3-2021.8)
1、screening and management of concomitant respiratory muscle failure: arterial blood gases
2、higher dose abatacept (≈20mg/kg, 3 doses within the first 2 weeks of treatment start) with dose adjustment
based on real‐time assessment of CD86 RO targeting peak value of CD86 RO≥80% within
72hours of abatacept and residual CD86 RO≥50% until resolution of myotoxicity severity to grade≤2.
3、addition of ruxolitinib + decrease in corticosteroids dose
4、asymptomatic cases: monitored after withholding ICI with no systematic immunossupressant treatment
JAK‐STAT pathway translational studies: RNA‐seq: ICI‐myocarditis mice and patients (database)
研究结果
Flow chart and evolution of the mortality of patients admitted for suspicion of ICI-myocarditis.

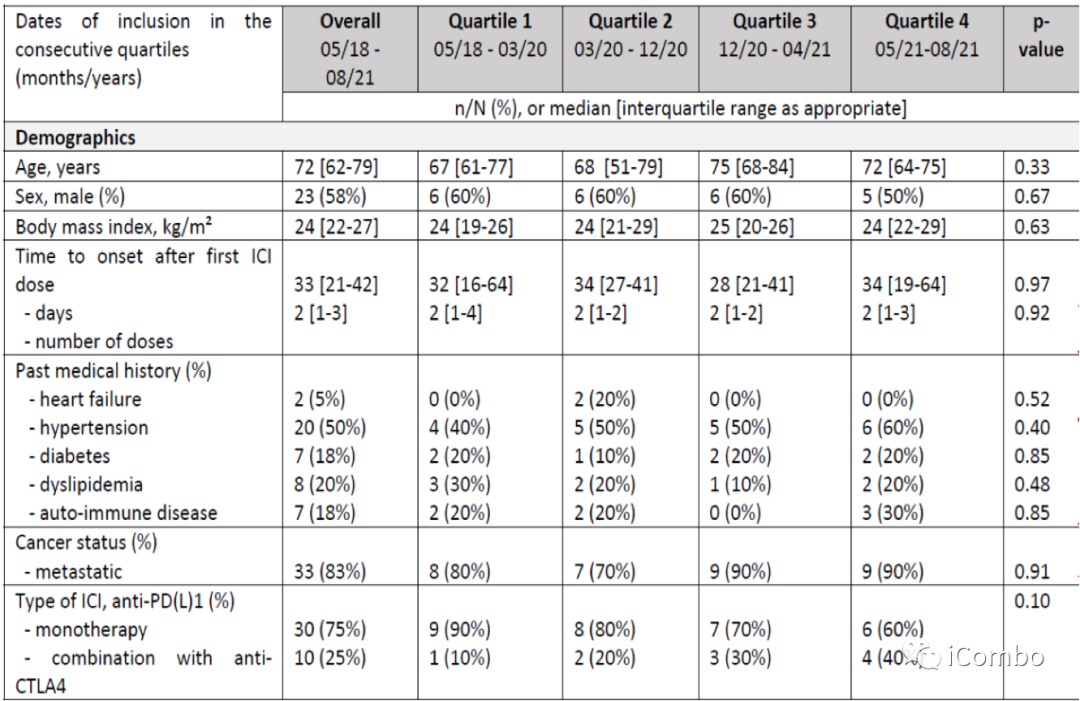
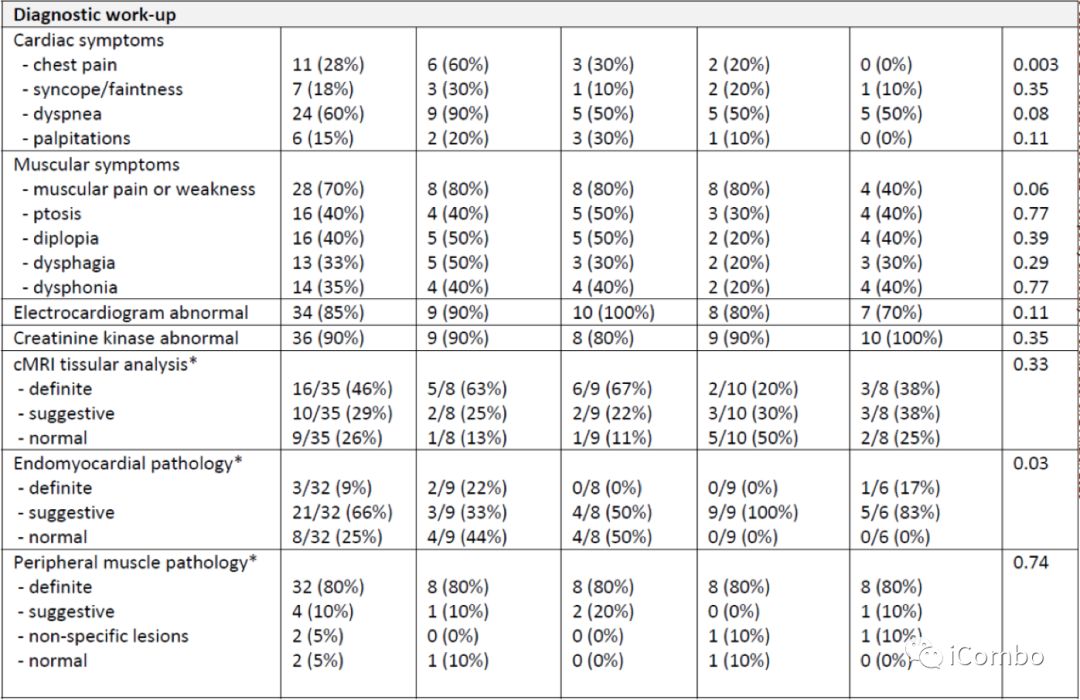
Characteristics of patients
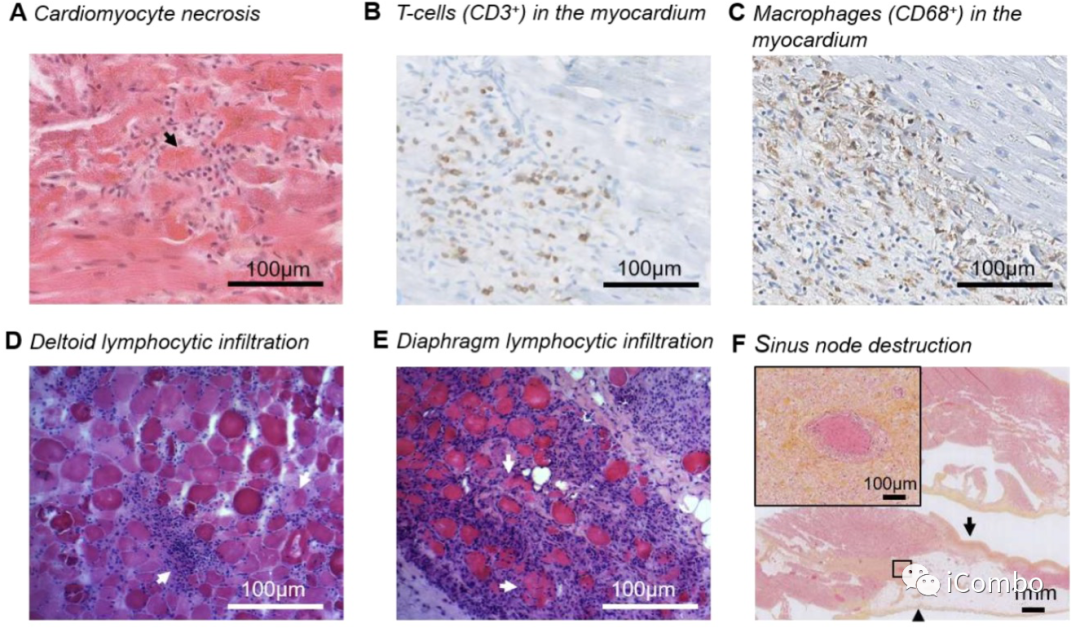
Examples of cardiac and skeletal muscle pathology findings on autopsies of 2 patients
Characteristics of patients

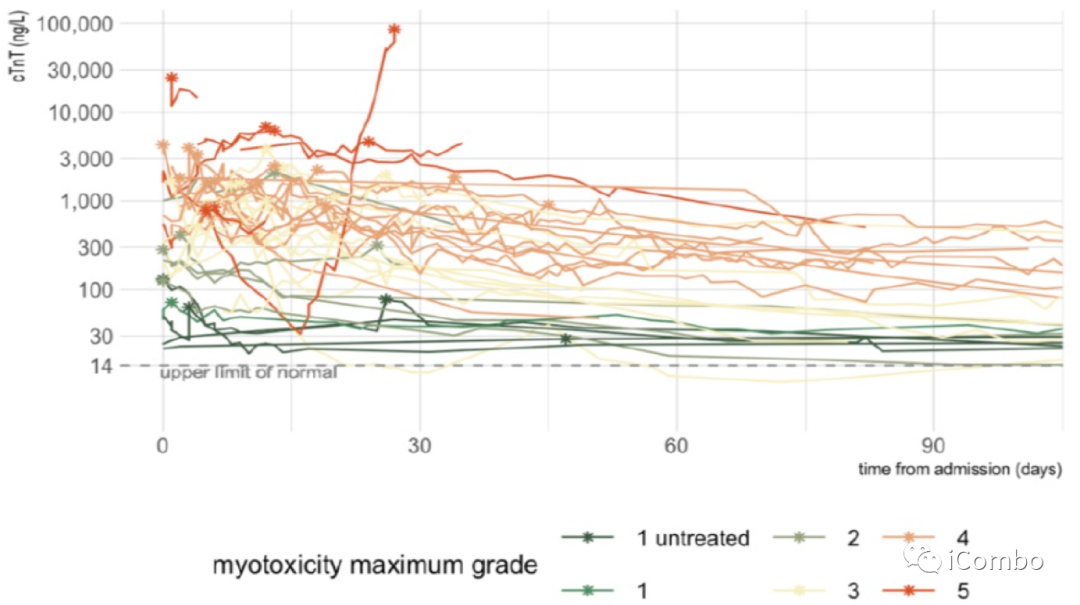
Troponin-T circulating levels as a function of ICI-myotoxicity secerity.
Effects of plasmapheresis on ICI circulating levels and immune checkpoint expression on peripheral blood mononuclear cells.
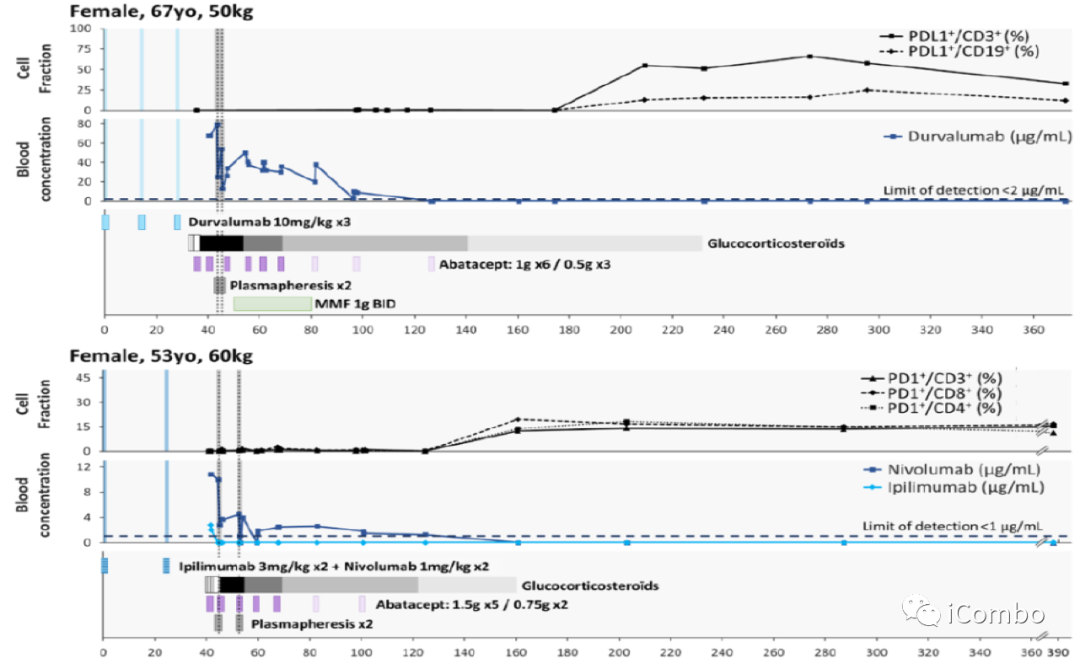
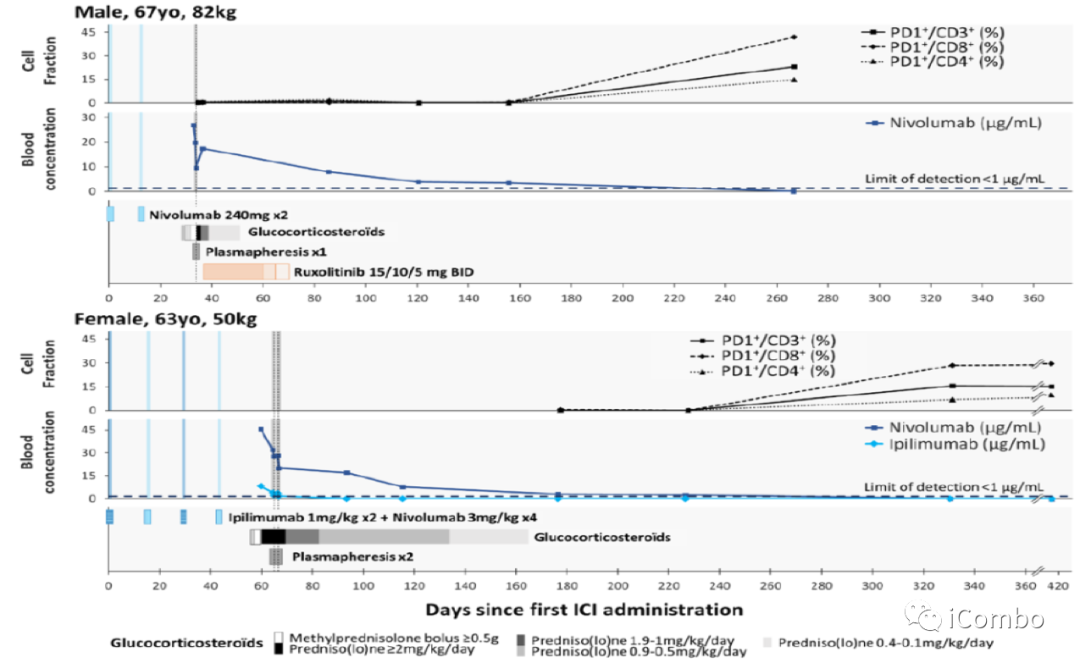
IC50:
~0.3 μg/mL for nivolumab ~0.02μg/mL for durvalumab
Abatacept binds its target CD86 on circulating monocytes dose-dependently.

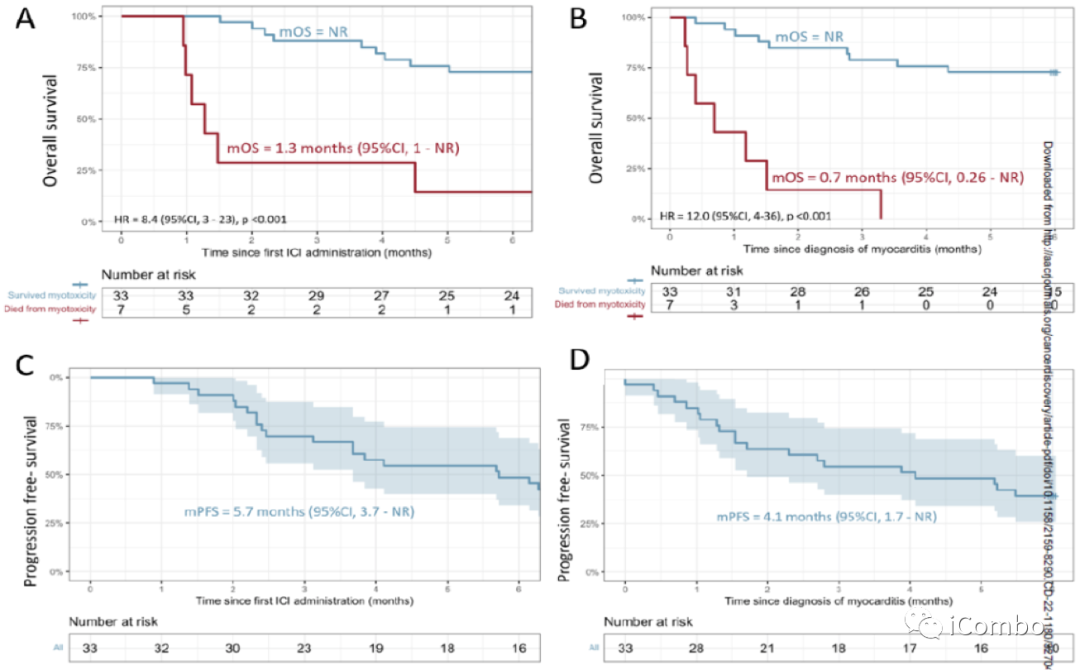
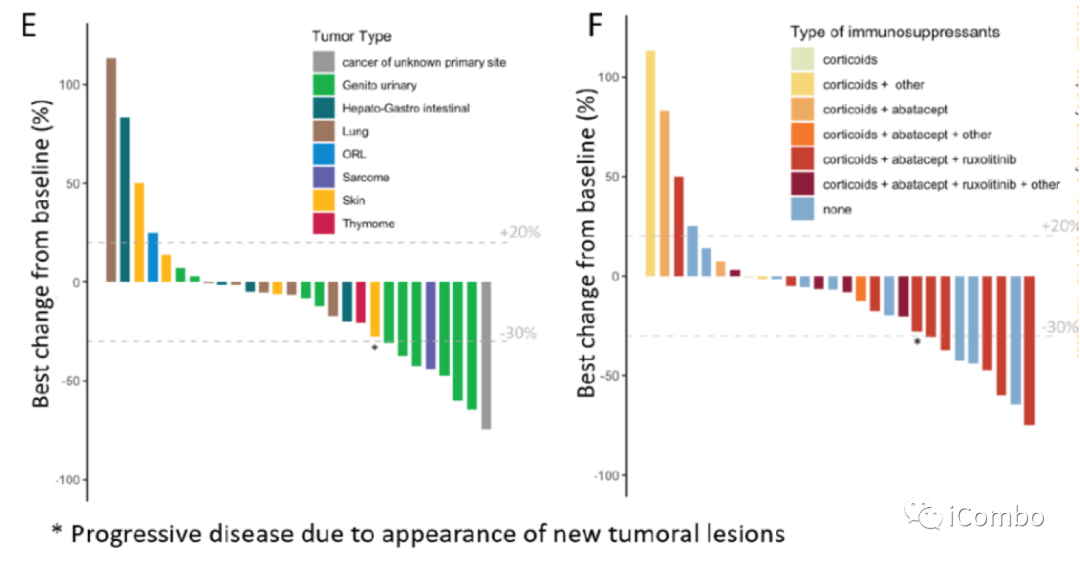
OS and cancer related outcomes in the cohort.
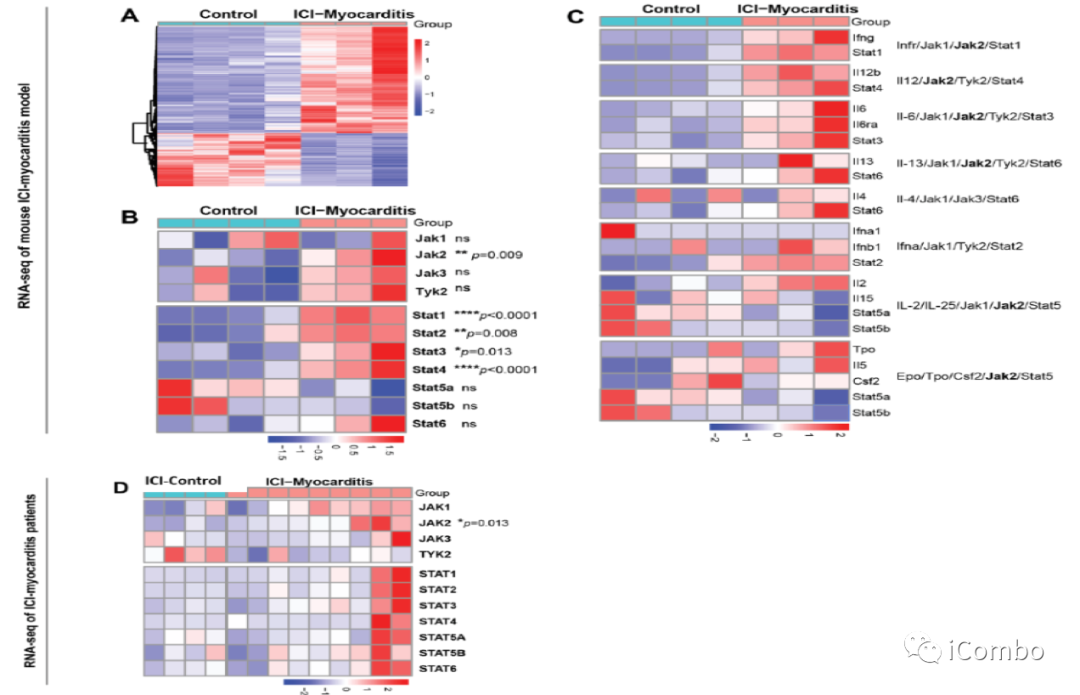
JAK-STAT pathway,particularly JAK2 is upregulated in ICI-myocarditis mice and patients.
OS and cancer related outcomes in the cohort.
研究总结
The authors showed that prompt high‐dose abatacept with real‐time assessment of CD86 RO immuno‐monitoring combined with ruxolitinib, corticosteroids, and active management of respiratory muscle involvement was associated with a decrease in ICI‐myotoxicity related death among ICI myocarditis patients.
One key element for improved care of ICI‐myocarditis has been our recognition that skeletal muscle involvement is near‐universal.
Limitations: non‐randomized observational study, small patients size, RNA seq on a small number of animals.
A prospective ongoing trial (NCT05195645)is in progress to assess the optimal drug mix, dosage and duration to be used to preserve ICI therapeutic effect while treating a severe irAE.
本网站所有内容来源注明为“梅斯医学”或“MedSci原创”的文字、图片和音视频资料,版权均属于梅斯医学所有。非经授权,任何媒体、网站或个人不得转载,授权转载时须注明来源为“梅斯医学”。其它来源的文章系转载文章,或“梅斯号”自媒体发布的文章,仅系出于传递更多信息之目的,本站仅负责审核内容合规,其内容不代表本站立场,本站不负责内容的准确性和版权。如果存在侵权、或不希望被转载的媒体或个人可与我们联系,我们将立即进行删除处理。
在此留言













#阿巴西普# #鲁索替尼# #免疫相关心肌炎#
23